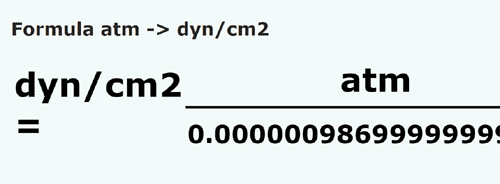
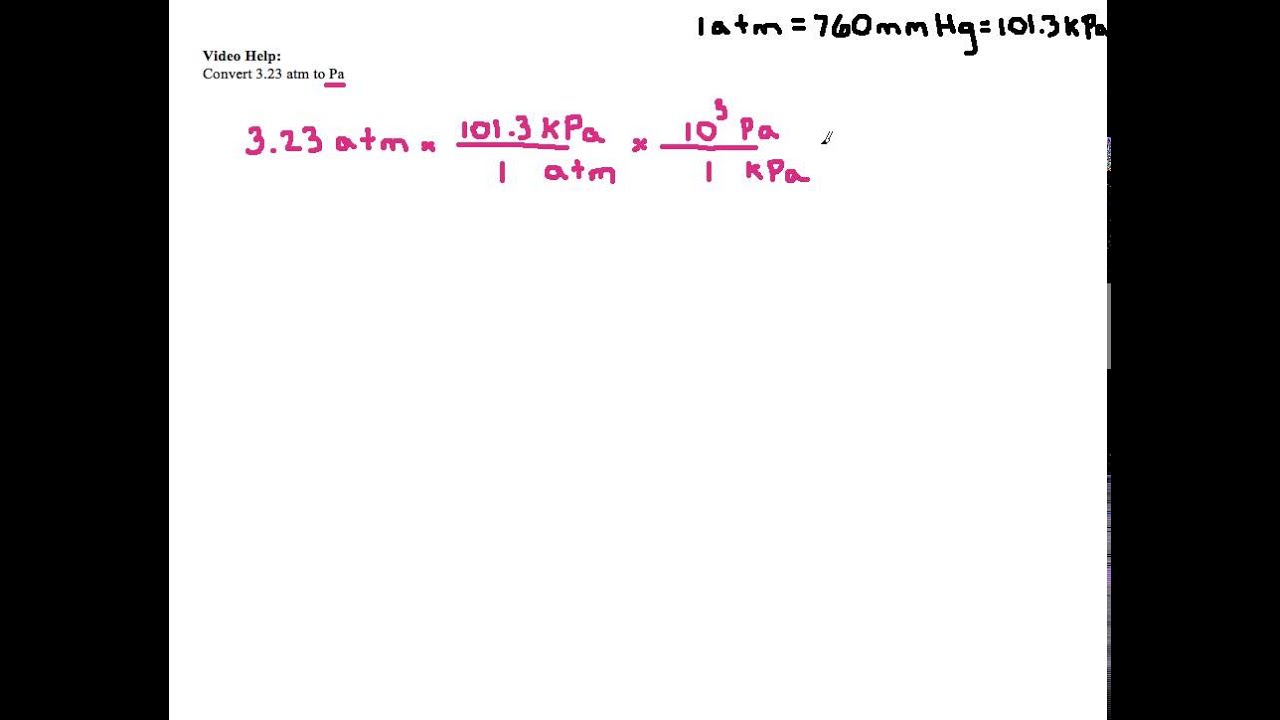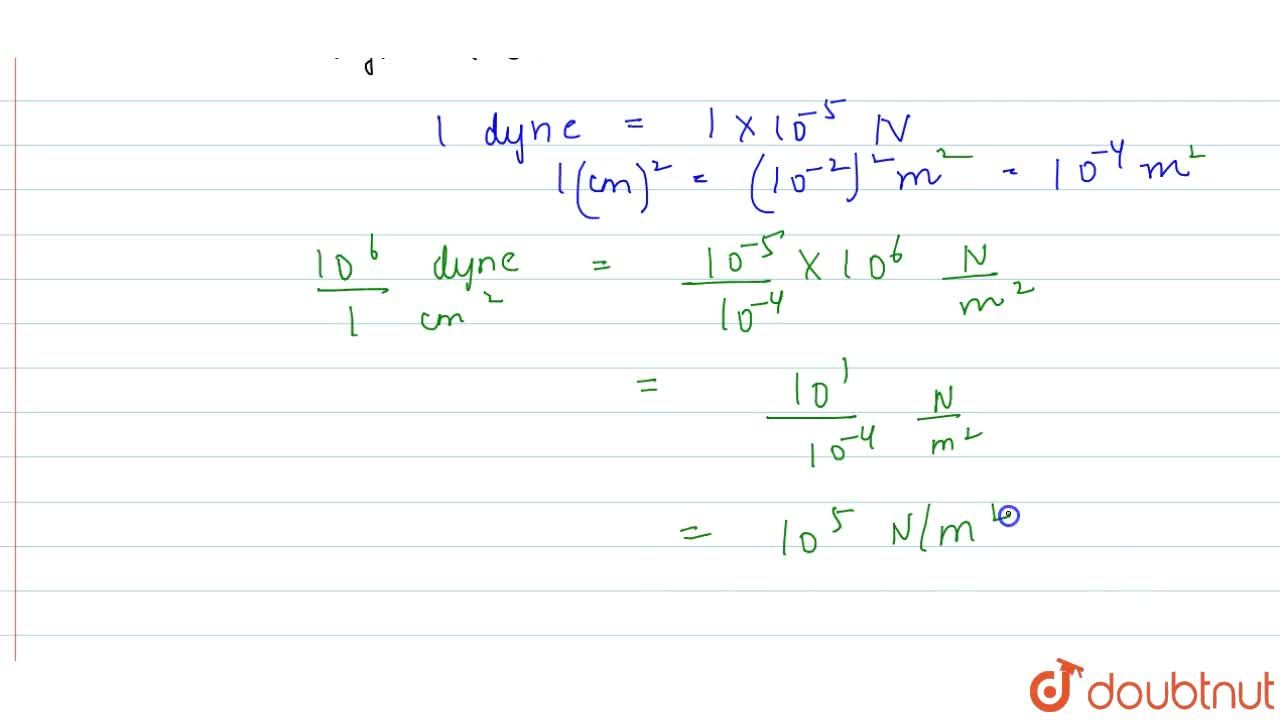convert a pressure of one atmospheric(=105 N m2)into dyne cm-2 could anyone please explain with detailed steps - Physics - Units And Measurements - 4719700 | Meritnation.com
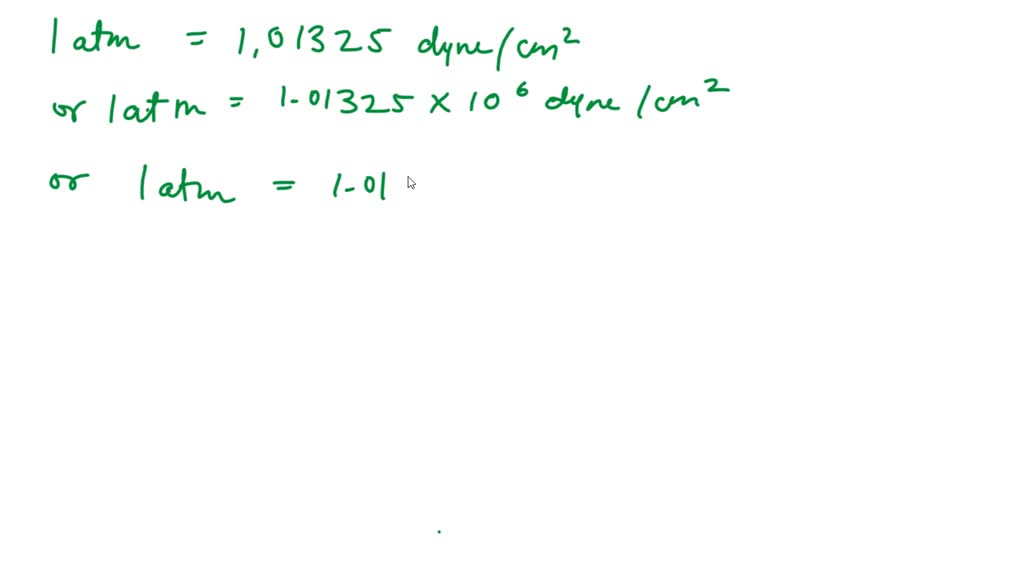
SOLVED: pts Question 1 Express an osmotic pressure of 7 atm in units of ' dyne/cm?. Using scientific nolation, the osmotic pressure is A x 108 dyne/cm?, Sclect | Solect pts

The value of atmosphere pressure is 10^(6) dynes/cm^(2). Find the value in S.I. using dimensional analysis?